Ответы 3
-
Ой, там 11 еще...Не заметил...ща допишу...
-
Автор:
gamsovbf - 6 лет назад
-
0
-
-
хоспаде спасибо огромное
-
Автор:
mangojoza - 6 лет назад
-
0
-
-
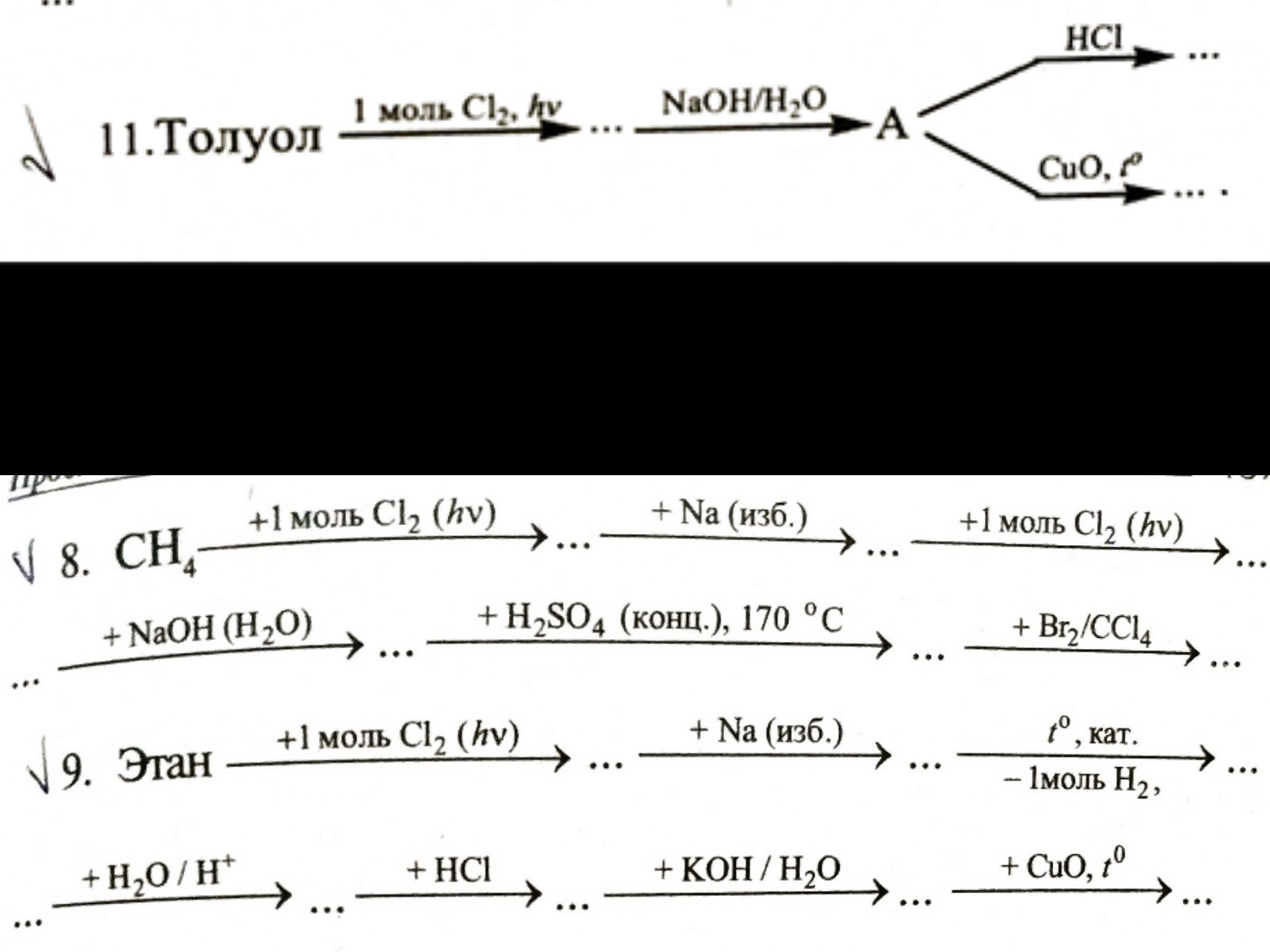
8. hvCH4 + Cl2 ------> CH3 - Cl + HClH3C - Cl + 2Na + Cl - CH3 ----> H3C - CH3 + 2NaCl hvH3C - CH3 + Cl2 -------> H3C - CH2 | ClH3C - CH2 + NaOH ------> H3C - CH2 + NaCl | (водн.) | Cl OH H2SO4 (конц.) 170°H3C - CH2 -------------------> H2C = CH2 + H2O | OH CCl4H2C = CH2 + Br2 ----------> H2C - CH2 | | Br Br9. hvH3C - CH3 + Cl2 -----> H3C - CH2 + HCl | ClH3C - CH2 + 2Na + H2C - CH3 ---> H3C - CH2 - CH2 - CH3 + 2NaCl | | Cl Cl Pt, tH3C - CH2 - CH2 - CH3 ------------> H2C = CH - CH2 - CH3 + H2 H+H2C = CH - CH2 - CH3 + H2O --------------> H3C - CH - CH2 - CH3 | OHH3C - CH - CH2 - CH3 + HCl ---------> H3C - CH - CH2 - CH3 + H2O | | OH ClH3C - CH - CH2 - CH3 + KOH ---------> H3C - CH - CH2 - CH3 + KCl | (водн.) | Cl OH O t //H3C - CH - CH2 - CH3 + CuO -----> H3C - CH2 - CH2 - C + Cu + H2O | \ OH H CH3 CH2 - Cl / \ hv / \| 0 | + Cl2 -------> | 0 | + HCl \ / \ / CH2 - Cl CH2 - OH / \ / \| 0 | + NaOH -------> | 0 | + NaCl \ / (водн.) \ / CH2 - OH CH2 - Cl / \ / \| 0 | + HCl ------> | 0 | + H2O \ / \ / O // CH2 - OH C -- H / \ t / \ | 0 | + CuO --------> | 0 | + Cu + H2O \ / \ /
-
Автор:
graciehsai - 6 лет назад
-
0
-
-
Добавить свой ответ
Еще вопросы
-
Спишите, выделенные слова разделите на слоги. Проверьте.
В школе.
Не опаздывай в школу. Не забывай дома нужные вещи. ЗАНИМАЙ своё место в классе до звонка. Слушай УЧИТЕЛЯ. Поднимай руку. Отвечай внятно.-
Предмет:
Русский язык -
Автор:
dahliarocha - 6 лет назад
-
Ответов:
3 -
Смотреть
-
-
√32(√2-√5)-3√10 вычислить
-
Предмет:
Алгебра -
Автор:
claudiachavez - 6 лет назад
-
Ответов:
1 -
Смотреть
-
-
площадь одной комнаты 35
[tex] {m}^{2} [/tex]
а площадь второй комнаты
[tex] \frac{5}{7} [/tex]
площадь первой комнаты ,Найдите площадь двух комнат.
помогите пожалуйста
-
Предмет:
Математика -
Автор:
timmydlln - 6 лет назад
-
Ответов:
2 -
Смотреть
-
-
Придумай рассказ о дереве которое ты считаешь чудесным где оно растёт? Как выглядит в разное время суток? при описании Используйте слова как, будто, словно.
Что бывает с деревом в разное время года? как относится к этому дереву? люди Почему ты считаешь его чудесным? Расскажи о добрых делах которые сделал ты или другие люди для растений.-
Предмет:
Литература -
Автор:
koko4 - 6 лет назад
-
Ответов:
1 -
Смотреть
-
How much to ban the user?
1 hour
1 day
100 years